La recherche sur les maladies négligées toujours en panne
Jean-Hervé Bradol
L'étude parue dans The Lancet, menée à l'initiative du DNDi (Drugs for Neglected Diseases Initiative) et à laquelle le Dr Jean-Hervé Bradol a participé, montre que seulement 4% des médicaments et des vaccins enregistrés entre 2000 et 2011 étaient destinés à lutter contre le paludisme, la tuberculose et les autres maladies négligées.
Summary
Background In 1975–99, only 1·1% of new therapeutic products had been developed for neglected diseases. Since then, several public and private initiatives have attempted to mitigate this imbalance. We analysed the research and development pipeline of drugs and vaccines for neglected diseases from 2000 to 2011.
Methods We searched databases of drug regulatory authorities, WHO, and clinical trial registries for entries made between Jan 1, 2000, and Dec 31, 2011. We defined neglected diseases as malaria, tuberculosis, diarrhoeal diseases, neglected tropical diseases (NTDs; WHO definition), and other diseases of poverty according to common definitions.
Findings Of the 850 new therapeutic products registered in 2000–11, 37 (4%) were indicated for neglected diseases, comprising 25 products with a new indication or formulation and eight vaccines or biological products. Only four new chemical entities were approved for neglected diseases (three for malaria, one for diarrhoeal disease), accounting for 1% of the 336 new chemical entities approved during the study period. Of 148 445 clinical trials registered in Dec 31, 2011, only 2016 (1%) were for neglected diseases.
Interpretation Our findings show a persistent insufficiency in drug and vaccine development for neglected diseases. Nevertheless, these and other data show a slight improvement during the past 12 years in new therapeutics development and registration. However, for many neglected diseases, new therapeutic products urgently need to be developed and delivered to improve control and potentially achieve elimination.
Funding None.
Introduction
Neglected diseases, understood broadly as diseases affecting populations in mainly low-income countries, are a leading cause of mortality, chronic disability, and povertyWHO. The global burden of disease: 2004 update. http://www.who. int/healthinfo/global_burden_disease/2004_report_update/en/ index.html (accessed on June 18, 2013).
Hotez PJ, Pecoul B. “Manifesto” for advancing the control and elimination of neglected tropical diseases. PLoS Negl Trop Dis 2010; 4: e718.
Beyrer C, Villar JC, Suwanvanichkij V, Singh S, Baral SD, Mills EJ. Neglected diseases, civil conflicts, and the right to health. Lancet 2007; 370: 619–627.. The need for greater political commitment to combat these diseases has been recognised by many World Health Assembly resolutions and the UN Millennium Development Goals.UN. Millennium development goals. We can end poverty 2015. http://www.un.org/millenniumgoals (accessed June 18, 2013).
Molyneux DH, Malecela MN. Neglected tropical diseases and the Millennium Development Goals—why the “other diseases” matter: reality versus rhetoric. Parasit Vectors 2011; 4: 234.
An analysis of drug development trends published in 2002 showed that only 1·1% of all drugs approved over the preceding 25 years (1975–99) were for neglected diseases,Trouiller P, Olliaro P, Torreele E, Orbinski J, Laing R, Ford N. Drug development for neglected diseases: a deficient market and a public-health policy failure. Lancet 2002; 359: 2188–94. despite these diseases accounting for 12% of the global health burden. The lack of appropriate research and development investment has been attributed to the absence of financial return for pharmaceutical and biotechnology companies, and a failure of public policy to establish sufficient enabling policies.Médecins Sans Frontières. Fatal imbalance: the crisis in research and development for drugs for neglected diseases. September, 2001. http://www.doctorswithoutborders.org/publications/reports/2001/ fatal_imbalance_short.pdf (accessed June 18, 2013).
Pécoul B, Chirac P, Trouiller P, Pinel J. Access to essential drugs in poor countries: a lost battle? JAMA 1999; 281: 361–67.
WHO. Research and development to meet health needs in developing countries: strengthening global financing and coordination. Report of the Consultative Expert Working Group on Research and Development: Financing and Coordination. http:// www.who.int/phi/cewg_report/en/index.html (accessed June 18, 2013).
We aimed to assess the state of research and development for neglected diseases over the past 12 years compared with other diseases.
Methods
Definition of neglected disease
Several definitions of neglected diseases are in common use. For this analysis, we used a broad definition, combining all terms from the following sources: the WHO list of neglected tropical diseases (NTDs),WHO. Neglected tropical diseases: diseases covered by NTD Department. http://www.who.int/neglected_diseases/diseases/en/ (accessed June 18, 2013). papers by Trouiller and colleaguesTrouiller P, Olliaro P, Torreele E, Orbinski J, Laing R, Ford N. Drug development for neglected diseases: a deficient market and a public-health policy failure. Lancet 2002; 359: 2188–94. and Hotez and colleagues,Hotez PJ, Fenwick A, Savioli L, Molyneux D. Rescuing the bottom billion through control of neglected tropical diseases. Lancet 2009; 373: 1570–75. PLoS Neglected Tropical Diseases journal disease scope,PLoS Neglected Tropical Diseases. PLoS Neglected Tropical Diseases Journal Scope. http://www.plosntds.org/static/scope. action (accessed June 18, 2013). G-FINDER 2011 report,Moran M, Guzman J, Abela-Oversteegen L, et al. Neglected disease research and development: Is innovation under threat? Policy Cures report, December, 2011. http://policycures.org/downloads/g-finder_2011.pdf (accessed June 18, 2013). and the BIO Ventures for Global Health 2012 report.BIO Ventures for Global Health. Developing new drugs and vaccines for neglected diseases of the poor: the product developer landscape. http://www.bvgh.org/LinkClick.aspx?fileticket=h6a0cJK9 drg%3d&tabid=91 (accessed June 18, 2013).
We included all diseases mentioned at least once in these sources, with the exception of several infections not uniquely endemic in low-income countries, including HIV/AIDS, meningitis, bacterial pneumonia, and rheumatic fever. We identified 49 neglected diseases, separating them into five categories: malaria, tuberculosis, diarrhoeal diseases, NTDs (WHO list of 17 NTDs),PLoS Neglected Tropical Diseases. PLoS Neglected Tropical Diseases Journal Scope. http://www.plosntds.org/static/scope. action (accessed June 18, 2013). or other neglected diseases (list of 19 not fitting into the other categories). The complete list of diseases is given in the appendix.
Approved products analysis
We identified newly approved drug and vaccine products by searching the European Medicines AgencyEuropean Medicines Agency (EMA). Human medicines. European public assessment reports. http://www.ema.europa.eu/ema/index. jsp?curl=pages/medicines/landing/epar_search. jsp&mid=WC0b01ac058001d124 (accessed June 18, 2013). and the US Food and Drug Administration (FDA) databases.Food and Drug Administration (FDA). Drugs@FDA. FDA Approved Drug Products. Advanced Search Options. http://www. accessdata.fda.gov/scripts/cder/drugsatfda/index. cfm?fuseaction=Search.Addlsearch_drug_name (accessed June 18, 2013). We included all products approved across all indications from Jan 1, 2000, to Dec 31, 2011. For neglected diseases, we also searched regulatory databases of the following countries, which are all signatories of the Pharmaceutical Inspection Convention and Pharmaceutical Inspection Co-operation Scheme:The Pharmaceutical Inspection Convention and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Members & Partners. Participating Authorities. http://www.picscheme.org/members.php (accessed June 18, 2013). European Union member states (Belgium, Czech Republic, Denmark, Estonia, France, Germany, Greece, Hungary, Portugal, Spain, Sweden, The Netherlands, and the UK), Canada, Australia, Switzerland, Iceland, and Argentina, and the regulatory databases of Japan, India, and Brazil. We used the earliest date of product approval listed in any of the above databases. We also searched for new products included on the WHO List of Prequalified Medicinal ProductsWHO. WHO list of prequalifi ed medicinal products. http://apps. who.int/prequal/query/ProductRegistry.aspx?list=all (accessed June 18, 2013). and the WHO Model List of Essential Medicines (Essential Medicines List).WHO. WHO model list of essential medicines. http://www.who. int/medicines/publications/essentialmedicines/en/ (accessed June 18, 2013).
Neglected diseases
For each registered product, we identified the marketing authorisation holder for the application and for the first submission in the regulatory database. For prequalified products, this information was available in the WHO database. This information was not available for products on the WHO Essential Medicines List because the international non-proprietary name, and not the marketed name, of the product is used. For these cases, we used the name of the dossier applicant for the product. We assessed the medical benefit (or innovation) of a given product using inclusion on the WHO Essential Medicines List as a proxy measure.
Clinical trials analysis
We assessed the clinical development pipeline for neglected-disease therapeutics by examining the number of ongoing phase 1–3 clinical trials listed in the US National Institutes of Health (NIH) clinical trials database,National Institutes of Health. ClinicalTrials.gov. http://www. clinicaltrials.gov (accessed Dec 31, 2011). and the WHO registry of clinical trials,National Institutes of Health. ClinicalTrials.gov. http://www. clinicaltrials.gov (accessed Dec 31, 2011). between Sept 1, 1999 (when the database was launched), and Dec 31, 2011. We also recorded all trials that were ongoing during the month of December, 2011. In the NIH database analysis, we included interventional and open phase 1–3 clinical trials. We excluded phase 4 studies; trials categorised as closed, withdrawn, terminated, suspended, or completed; and trials in which the main indication was misclassified in the registry database. Additionally, for neglected diseases, we included studies classified as closed (not recruiting or enrolling by invitation only) but still active; we included interventional studies with no defined clinical-trial phase on a case-by- case basis (78 studies included). In the WHO registry, we included interventional and currently recruiting studies. Studies already listed in the NIH database, phase 4 studies, and trials in which the main indication was misclassified were excluded. Recruitment status was unknown for 23 interventional studies, and these studies were not included in the analysis.
For each clinical trial, product type was classified as for approved products (NCE, new indication, new formulation, fixed-dose combination, or vaccine or biological product). Additionally, when approved drugs were combined for a neglected-disease indication but not manufactured as a fixed-dose combination, we classified them as a new association of registered drugs.
We classified clinical trial sponsors into four categories: public organisations (including government, academic, and public research institutes); private not-for-profit organisations (including product development partnerships, charities, foundations, and philanthropic institutions); private industry, for-profit entities (including pharmaceutical and biotechnology companies); and a mixed-sponsor category when products were studied in clinical trials by different categories of sponsors.
Finally, for each neglected disease, we assessed the existence of a vaccine or other intervention to prevent the infection. After assessing new products in development in 2011, we classified the neglected diseases into three categories of research and development need: diseases with no crucial clinical gaps in research and development (those for which either preventive measures or at least two field-adapted, safe, effective treatments to respond to drug resistance exist); diseases with crucial gaps in research and development and with ongoing clinical research; and diseases with crucial gaps in research and development but with no ongoing clinical research.
Role of the funding source
There was no funding source for this study. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results
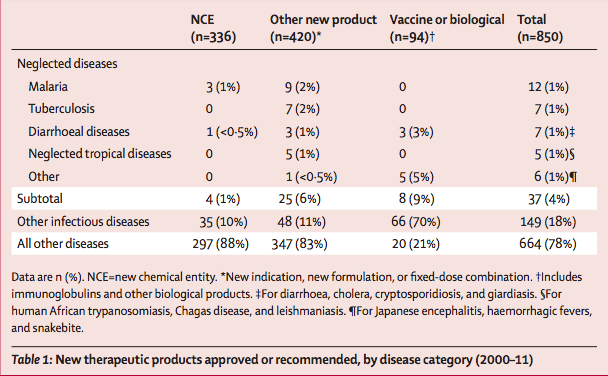
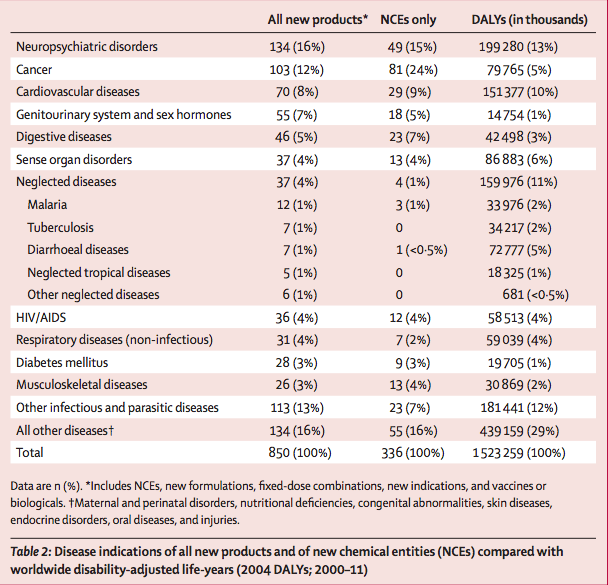
850 new therapeutic products (NCEs, new indications, new formulations, fixed-dose combinations, and vaccines and biologicals) were approved by different regulatory bodies between Jan 1, 2000, and Dec 31, 2011. Of these, 37 (4%) were for neglected diseases, including 29 drugs and eight vaccines (table 1). Collectively, neglected diseases accounted for about a tenth of the global disease burden in 2004 (table 2).
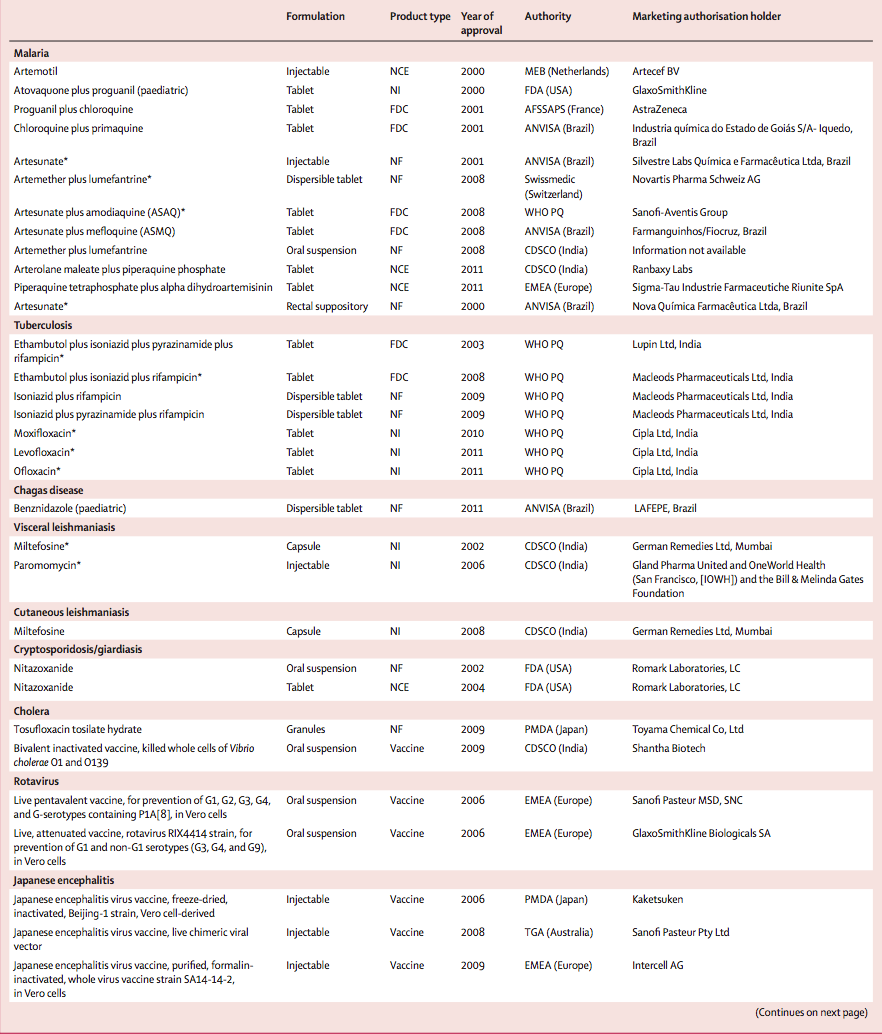
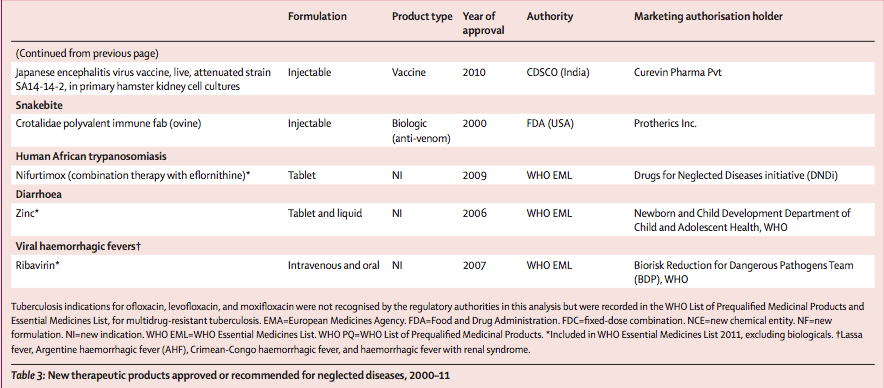
Of the 336 NCEs approved between 2000 and 2011, only four (1%) were for neglected diseases. Among the other product types, 25 (6%) of 420 new indications, new formulations, and fixed-dose combinations, and eight (9%) of 94 vaccines and biologicals, were for neglected diseases. Of these 37 new products approved for neglected diseases, most were for malaria (n=12), including three of the four NCEs (table 3). The other NCE was indicated for diarrhoeal disease (cryptosporidiosis or giardiasis). No NCE was developed in 2000–11 for tuberculosis or any NTDs. The vaccines and biologicals approved were for Japanese encephalitis (n=4) and diarrhoeal diseases (n=3), with one antivenom developed for snakebite.
23 (7%) NCEs were approved for other infectious and parasitic diseases, which accounted for about 12% of total disease burden (table 2). By contrast with those for infectious diseases, 24% of all NCEs were indicated for cancer, which accounted for about 5% of the global disease burden. The next largest categories of NCE approvals were for neuropsychiatric disorders and cardiovascular diseases (table 2).
In terms of medical benefit, 14 (48%) of the 29 new products (excluding vaccines and biologicals) approved for neglected diseases in 2000–11 were included in the WHO Essential Medicines List, by contrast with only 26 (4%) of 727 for all other diseases. 30 (81%) of 37 new product registrations were filed by regional generic or public pharmaceutical companies, including 12 by Indian manufacturers. Seven product registrations were filed by large, private pharmaceutical companies.
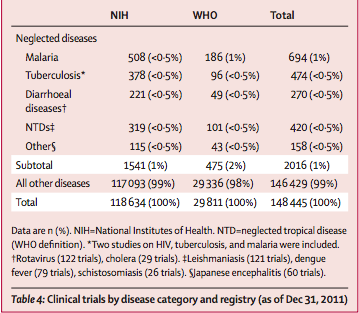
Between Sept 1, 1999, and Dec 31, 2011, 148 445 clinical trials for all diseases were registered in the NIH and WHO databases, only 2016 (1%) of which were for neglected diseases (table 4). As of Dec 31, 2011, we identified 383 active clinical trials for neglected diseases—malaria and tuberculosis accounted for more than half these trials (table 5).
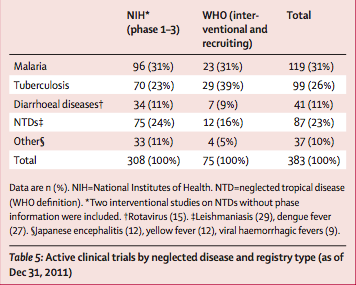
Of the 383 active clinical trials for neglected diseases, 122 (32%) are testing a new vaccine candidate or biological product; 57 (15%) are testing a new formulation (including fixed-dose combinations), indication, or association; and 24 (6%) are testing an NCE. The remainder (180 studies) are directed at follow-up studies or testing new dosages or alternative regimens (n=43); an approved drug for geographical extension (n=32); dietary supplements and adjunct therapies (n=23); drugs to treat disease complications (n=16); studies of subpopulations with HIV (n=16); prevention interventions (n=12); studies in pregnancy (n=11); diagnostics (n=8), or other procedures (n=8); challenge studies (n=7); or pharmacological studies (n=4).
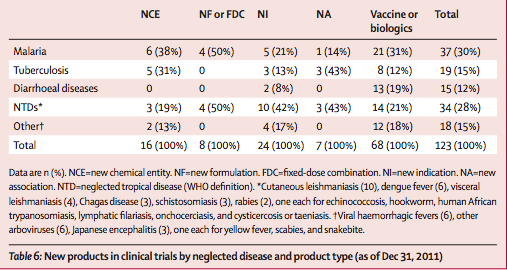
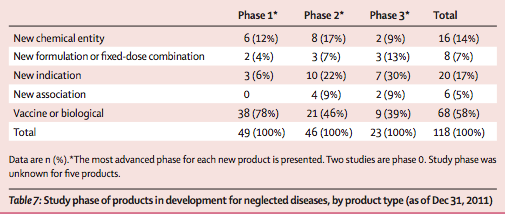
123 new products are being tested in these 383 active clinical trials for neglected diseases (table 6). Most products in development are vaccines and biological products (table 6). Slightly more than a quarter of products in development are for NTDs, but only three are NCEs (for onchocerciasis, Chagas disease, and human African trypanosomiasis). Two other NCEs are in development for other neglected diseases (Ebola and Marburg viruses). Vaccine (and biological-product) candidates are in less advanced stages of clinical development compared with other products, with most still in phase 1 (table 7). Of the 34 products that are new formulations (including fixed- dose combinations), new indications, or new associations, most (29 [85%]) are in phase 2 or 3 clinical trials. Of the 16 NCEs in development, half are in phase 2 and two are in phase 3.
Of the 49 diseases included in this study (appendix), only 11 had no crucial research gaps (eg, dracunculiasis). For 25 diseases, research and development gaps exist and clinical research is ongoing to address some of these gaps (eg, for tuberculosis, human African trypanosomiasis, filariasis, snakebite). For 13 (27%) diseases, gaps exist but no clinical research was ongoing at the time of analysis (eg, Buruli ulcer, mycetoma, food-borne trematode infections). Assessment of study sponsorship, as listed in the NIH and WHO clinical trials registries, for the 123 products in development showed the involvement of public organ- isations in 66 products (54%), private industry in 28 products (23%), and private not-for-profit organisations in 19 products (15%), with the remaining 10 products (8%) involving a mix of sponsors. About half of all new products in development sponsored by private not-for- profit organisations are for NTDs (ten of 19), and the other half are for malaria, tuberculosis, and diarrhoeal diseases (nine of 19). All three NCEs for NTDs are being sponsored by private not-for-profit organisations.
|
Panel: Research in context Systematic review We searched PubMed for publications using the terms “neglected diseases” and “global burden of disease” from Jan 1, 1999, to Dec 31, 2011. We searched the publications and websites of WHO and organisations involved in neglected-diseases research, financing, and analysis (BIO Ventures for Global Health, Policy Cures, Global Alliance for TB Drug Development, Medicines for Malaria Venture [MMV], PATH, Drugs for Neglected Diseases initiative [DNDi]). Attrition rates applied for forecasting future product approvals came from two research articlesKola I, Landis J. Can the pharmaceutical industry reduce attrition rates? Nat Rev Drug Discov 2004; 3: 711–15. 31 Nwaka S, Ridley RG. Virtual drug discovery and development for neglected diseases through public-private partnerships. Nat Rev Drug Discov 2003; 2: 919–28. Interpretation The results of our analysis update the current evidence on therapeutic product approvals for neglected diseases through 2011, and provide future approval forecasts for the next decade, based on highly relevant disease scope. Our findings are consistent with those reported in similar studies,Cohen J, Dibner MS, Wilson A. development of and access to products for neglected diseases. PLoS One 2010; 5: e10610. Despite positive advances over the past decade in therapeutics for neglected diseases, a persistent gap remains in product development for most of the neglected diseases. Therapeutic advancements have for the most part been achieved through repurposed or reformulated versions of existing drugs. A major research and development gap remains in new chemical entities for neglected diseases, both in terms of recent approvals and ongoing clinical development. For many neglected diseases, new therapeutic products urgently need to be developed and delivered to improve disease control and potentially achieve elimination. |
Discussion
Despite substantial political attention towards the burden of neglected diseases, we detected no evidence of a substantial improvement in research and development activity compared with previous decades. Some progress has been made, but these advancements have not in large part redressed the research and development imbalance, reported more than a decade ago, in truly new therapeutic products for neglected diseases (panel). Most new products are repurposed versions of existing products.
From 2000 to 2011, only 4% of all approved new products (a fraction of which are vaccines), and only 1% of all approved NCEs, were indicated for neglected diseases, although the global health burden of these diseases is estimated at 11%. Similarly, only 1% of all existing clinical trials in December, 2011, were for neglected diseases. Very few ongoing trials are assessing NCEs (6%), with greater emphasis on vaccines (32%) and repurposing of existing drugs (15%).
This study had several strengths and weaknesses. We applied a broad definition of neglected diseases in an attempt to provide a comprehensive picture. However, we could not reliably estimate the burden of several of the diseases included, because their burden might be underestimated by DALYs or because data were unavailable.Fenwick A. The global burden of neglected tropical diseases. Public Health 2012; 126: 233–36.
King CH, Dickman K, Tisch DJ. Reassessment of cost of chronic helminthic infection: a meta-analysis of disability-related outcomes in endemic schistosomaisis. Lancet 2005; 365: 1561–69. An analysis of 2010 global disease burden reported a similar DALY percentage for neglected diseases (10·5%), based on a slightly different list of diseases.Murray CJ, Vos T, Lozano R, et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012; 380: 2197–223. However, due to differing definitions of neglected diseases, we excluded some diseases of importance to low-income countries, such as bacterial pneumonia and meningitis, which could arguably be regarded as neglected diseases. Thus, MenAfriVac (Serum Institute of India Ltd, Pune, India), a new vaccine developed for group A meningitis endemic in sub- Saharan Africa, registered in India, and on the WHO List of Prequalified Medicinal Products, was not included.
Registration data were not openly available on the internet for some countries (eg, South Africa, China). China is not a member of the Pharmaceutical Inspection Co-operation Scheme. However, we think it unlikely that new products would be registered only in these countries and nowhere else. To cover all the neglected diseases in our analysis, we expanded the registration database search beyond only FDA and European Medicines Agency to other databases and registries of a large number of endemic and non-endemic countries.
Also, during the time period assessed (2000–11), the WHO prequalification process was created (in 2001) for drugs, especially those for HIV, tuberculosis, and malaria. We included new products on the WHO List of Prequalified Medicinal ProductsWHO. WHO list of prequalifi ed medicinal products. http://apps. who.int/prequal/query/ProductRegistry.aspx?list=all (accessed June 18, 2013) and WHO Essential Medicines ListWHO. WHO model list of essential medicines. http://www.who. int/medicines/publications/essentialmedicines/en/ (accessed June 18, 2013). to account for neglected-disease research and development efforts not captured by our search of regulatory authorities. Consequently, WHO prequalification was the basis for identifying all seven of the new products for tuberculosis, and one for malaria, in 2000–11, and the Essential Medicines List accounted for the new indications for human African trypanosomiasis, diarrhoeal disease, and viral haemorrhagic fevers. However, certain loose combinations of products that are neither registered nor included on the WHO Essential Medicines List (eg, the combination of sodium stibogluconate and paromomycin for the treatment of visceral leishmaniasis in Africa) were not counted as new products.
Use of inclusion in the WHO Essential Medicines List as a proxy metric for medical innovation has its limits because the list favours infectious diseases affecting, and low-cost products for, low-income countries. Also, inclusion is based on voluntary application, which might result in the missed counting of any products for which the sponsor did not apply for inclusion on the list.
Findings from a 2002 analysis showed that between 1975 and 1999 only 16 (1·1%) of 1393 new products had been developed for neglected diseases.6 Another analysis of the same period published in 2010 but using different product-approval information sources showed double the number of new products developed (n=32).Cohen J, Dibner MS, Wilson A. development of and access to products for neglected diseases. PLoS One 2010; 5: e10610. For the period 2000–09, the same investigators reported a lower number of new products (26; 15 excluding HIV/AIDS, bacterial pneumonia, and meningitis) compared with our analysis. The report of 16 new products approved over the 25-year period of 1975–99 gives an average of 0·6 product per year, whereas the 2010 report of 32 new products over the same time period gives an average of 1·3 products per year. Our analysis showed that 29 new products, excluding vaccines and biologicals, were approved over the 12-year period of 2000–11, corresponding to an average of 2·4 products per year. Although direct comparison between the three studies is not possible because of differences in disease and product definitions and methods, their collective findings show a trend of a slight increase in products approved per year.
Malaria and tuberculosis accounted for 19 of the 29 new products approved for neglected diseases in 2000–11. Only five new products were developed for NTDs, all of which were for only three of the 17 NTDs as defined by WHO.WHO. Neglected tropical diseases: diseases covered by NTD Department. http://www.who.int/neglected_diseases/diseases/en/ (accessed June 18, 2013). Of the 29 new products, few are truly innovative: most are based on the repurposing of existing treatments, namely reformulations, new indications, or fixed-dose combinations. This approach, which is faster, cheaper, and less risky than novel drug discovery and development, has resulted in some important advancements for some diseases, as evidenced by the relatively high level of inclusion of new products on the WHO Essential Medicines List during 2000–11 (48%).
Only four NCEs have been developed for neglected diseases in the past 12 years. Of these, three were for malaria (of which two were derivatives of artemisinin, which was developed in the 1970s; the third combined an artemisinin derivative with an existing drug), and the fourth NCE, nitazoxanide, was first developed for cryptosporidiosis, an HIV-related opportunistic infection, and was later shown to be effective against giardiasis. Apart from this one NCE, no new drug class for neglected diseases has been approved in the past 12 years. The deficiency in research and development for neglected diseases seen in our study has also been reported in the area of antibiotics, where new drugs are urgently needed to treat drug-resistant bacterial infections.Spellberg B, Powers JH, Brass EP, Miller LG, Edwards JE. Trends in antimicrobial drug development: Implications for the future. Clin Infect Dis 2004; 38: 1279–86.
Jabes D. The antibiotic R&D pipeline: an update. Curr Opin Microbiol 2011; 14: 564–69.
Seven of the marketing authorisations for products approved in 2000–11 were granted to large European pharmaceutical companies. This appears to represent a change compared to the situation over a decade ago, when in 2001 large pharmaceutical companies were reported to have spent less than 1% of their research and development budgets on neglected diseases over the previous fiscal year.7 European leadership was also reported in 2008 and is still present in the 2012 report of the Access to Medicine index.Access to Medicine Foundation. The access to medicine index 2012. http://www.accesstomedicineindex.org/sites/www. accesstomedicineindex.org/fi les/2012-access-to-medicine-index-fullreport-clickable.pdf (accessed June 18, 2013). Still, investments by the pharmaceutical industry have been limited to diseases with large volumes or a potential market, namely malaria, rotavirus, and Japanese encephalitis.
The development of innovative NCEs and biologicals, including vaccines, is a lengthy process, especially compared with the time taken to repurpose an existing drug. Recent investments and the efforts of product development partnerships and other partnerships have in large part followed the relatively easier approach of developing repurposed products rather than NCEs. However, several NCEs were in ongoing trials during the time of our analysis, but were not yet approved.
As of Dec 31, 2011, 123 new products were in the research pipeline. More than half (55%) were vaccines or biological products for a larger range of diseases than in the past. But NCEs remain poorly investigated, representing only 13% of the research efforts for neglected diseases. In terms of neglected-disease areas, malaria remains the primary focus of research and development. For NTDs, increased efforts are apparent: 28% of new products (34 of 123) in clinical development for NTDs, compared with 13% of new products (five of 37) approved in 2000–11. These 34 products in development were intended for 11 NTDs, whereas the five new products approved in 2000–11 covered only three NTDs. However, three NTDs (Buruli ulcer, food- borne trematode infections, and leprosy) and ten other neglected diseases remain excluded from any product development although new treatment or preventive interventions are needed.
Findings from a 2010 study showed 97 new products in clinical development as of July, 2009 (65 when excluding indications for HIV/AIDS, bacterial pneumonia, and meningitis), with the most being vaccines (46 of 65).Cohen J, Dibner MS, Wilson A. development of and access to products for neglected diseases. PLoS One 2010; 5: e10610. This finding is consistent with our findings, which covered a more recent period (until December, 2011). Another study looking at products developed for neglected diseases that could potentially be granted an FDA priority review voucher detected 62 new products in clinical development in July, 2011 (17 drugs and 45 vaccines), for 16 neglected diseases.Stefanakis R, Robertson AS, Ponder EL, Moree M. Analysis of neglected tropical disease drug and vaccine development pipelines to predict issuance of FDA priority review vouchers over the next decade. PLoS Negl Trop Dis 2012; 6: e1803. Despite differences in methodology, these findings also showed a high proportion of vaccine candidates in development. Unsurprisingly, clinical trials investigating new indications of existing drugs were at a more advanced stage than vaccine trials, which are mostly in phase 1 or NCE trials, which are mostly phases 1–2.
The predicted number of new products (excluding vaccines and biologicals) for neglected diseases to be approved in the coming years can be estimated by extrapolating the number of products in phase 1–3 clinical development and applying an attrition rate for infectious diseases.Kola I, Landis J. Can the pharmaceutical industry reduce attrition rates? Nat Rev Drug Discov 2004; 3: 711–15.
Nwaka S, Ridley RG. Virtual drug discovery and development for neglected diseases through public-private partnerships. Nat Rev Drug Discov 2003; 2: 919–28.
Drugs for Neglected Diseases initiative. 2011–2018 business plan. http://www.dndi.org/images/stories/pdf_aboutDNDi/ BusinessPlanWebSmall.pdf (accessed June 18, 2013). Success rates applied were 70% for NCEs in phase 1, 50% for those in phase 2, and 65% for those in phase 3, and 80% for all phases for new indications, associations, and formulations, except 70% for phase 1 and 85% for phase 3 of new formulations. Predicted registration success rates were 95% for NCEs and 100% for other products. We forecast that the current pipeline has the potential to deliver about 28 new registered products over the next 6 years, but only five NCEs. This forecasted estimate corresponds to an average of 4·7 new products per year through 2018, which represents a considerable acceleration compared with the finding of an average of 2·4 new products per year developed in 2000–11.
For vaccines and biologicals, 15 new products could be expected in the next 10 years applying a conservative 22% success rate,Serdobova I, Kieny MP. Assembling a global vaccine development pipeline for infectious diseases in the developing world. Am J Public Health 2006; 96: 1554–59. again suggesting an accelerating trend in research and development. Vaccine development generally has higher attrition rates and longer timelines than does drug development, and early clinical trials for vaccines cannot be compared directly with early clinical trials for drugs when predicting approvals. Another assessment of NTD drug and vaccine approvals predicted lower numbers than our forecast of new products in the next 5 years, though this prediction is based on more specific criteria, including time to registration using a specific FDA-approval pathway for neglected diseases, and narrower disease scope.Stefanakis R, Robertson AS, Ponder EL, Moree M. Analysis of neglected tropical disease drug and vaccine development pipelines to predict issuance of FDA priority review vouchers over the next decade. PLoS Negl Trop Dis 2012; 6: e1803.
Ongoing clinical research will probably bring new treatment options for malaria and tuberculosis, with several NCEs in the pipeline, but is not likely to produce a substantial number of NCEs for other neglected diseases. Even if vaccine development accelerates and offers potential solutions in the future, the overall health burden of neglected diseases will not be resolved. Vaccines will probably not be developed for every neglected disease, and in the short term, patients with infections and at risk of death or serious disability will require immediate treatment. The spread of resistance to infectious and parasitic organisms, notably to malaria, tuberculosis, and diarrhoeal infections, and the toxicity and burdensome administration of some treatments, still justify the development of new products for most neglected diseases.
The anticipated near-doubling of new products in the coming years compared with previous years is the result of substantial changes to the research and development landscape over the past 15 years. New consortia, partnerships, and business models for drug discovery have emerged, including product development partnerships, which have attempted to fill research and development gaps and catalyse new efforts for several neglected diseases. With the support of new private and public funding, including important contributions by the Bill & Melinda Gates Foundation since 2000, there has been an increase of resources allocated to innovation for neglected diseases, reaching US$3 billion in 2011.Moran M, Guzman J, Henderson K, et al. Neglected disease research and development: a fi ve year review. G-FINDER 2012. Policy Cures. http://www.policycures.org/g-fi nder2012.html (accessed June 18, 2013). Nevertheless, in 2010 only 1% of research and development investment for global health was allocated to neglected diseases.Røttingen JA, Regmi S, Eide M, et al. Mapping of available health research and development data: what’s there, what’s missing, and what role is there for a global observatory? Lancet 2013; published online May 20, 2013. http://dx.doi.org/10.1016/S0140-6736(13)61046-6.
Although the public sector provides two-thirds of all research and development funding for neglected diseases, at nearly $2 billion per year, government funding is shifting away from product development and towards traditional basic research.Moran M, Guzman J, Henderson K, et al. Neglected disease research and development: a fi ve year review. G-FINDER 2012. Policy Cures. http://www.policycures.org/g-fi nder2012.html (accessed June 18, 2013). Additionally, funding by industry and philanthropy, the sectors that customarily fund product development, is dropping or refocusing on only a few products and diseases, namely dengue fever, tuberculosis, and bacterial pneumonia and meningitis.Moran M, Guzman J, Henderson K, et al. Neglected disease research and development: a fi ve year review. G-FINDER 2012. Policy Cures. http://www.policycures.org/g-fi nder2012.html (accessed June 18, 2013). Our analysis shows that public organisations have a leading role as the main sponsor for more than half (54%) of all products in clinical development for neglected diseases. For NTD drug development, public institutions and private not-for-profit organisations play an important part as product clinical-trial sponsors, and for vaccine development, the pharmaceutical industry is the leading sponsor. Private industry sponsors nearly a quarter (23%) of new product trials for neglected diseases, compared with a near standstill in product development a decade ago.Hotez PJ, Pecoul B. “Manifesto” for advancing the control and elimination of neglected tropical diseases. PLoS Negl Trop Dis 2010; 4: e718
Nwaka S, Ridley RG. Virtual drug discovery and development for neglected diseases through public-private partnerships. Nat Rev Drug Discov 2003; 2: 919–28. However, we were not able to assess all partnerships and collaborations involved in the development of new products for neglected diseases.
Our findings show a persistent deficiency in product development for neglected diseases, although in the past 12 years positive advances have been seen for neglected- disease treatments, based mainly on the number of newly approved drug reformulations, repurposed products, and vaccines, as well as the number of ongoing clinical trials, especially for vaccines. Nevertheless, a major research and development gap remains in NCEs for neglected diseases, both in terms of new approvals and ongoing clinical development as shown by only 1% of existing clinical trials focused on this area. Malaria, tuberculosis, and diarrhoeal diseases remain the main focus of product- development research, with little focus on other neglected diseases. Providing the required treatments to control and then eliminate neglected diseases is a crucial concern and will require investment efforts into research and development for neglected diseases on all fronts.
Contributors
NS-W, BPe, BPé, and J-HB had the idea for and designed the study. BPe and CS collected and compiled the data. BPe, NS-W, PO, PT, NF, BPé, and J-HB did the data analysis and interpretation. BPe wrote the first draft of the paper with input from all other authors. All authors read and approved the final paper.
Conflicts of interest
We declare that we have no conflicts of interest.
Acknowledgments
PO is a staff member of WHO. The opinions and views expressed in this paper are those of the authors, and do not necessarily represent the decisions, policy, or views of WHO or the Centre Hospitalier Universitaire Bordeaux. We thank the following individuals for their invaluable expertise and assistance: Cherise P Scott, Alejandro Costa, Carla Botting, Peter J Hotez, Farrokh Modabber,
Jean-François Alesandrini, Graciela Diap, Gabriela Chaves, Pascale Boulet, Bruno Scherrer, and Oliver Yun.
Pour citer ce contenu :
Jean-Hervé Bradol, « La recherche sur les maladies négligées toujours en panne », 30 octobre 2013, URL : https://msf-crash.org/fr/medecine-et-sante-publique/la-recherche-sur-les-maladies-negligees-toujours-en-panne
Si vous souhaitez critiquer ou développer cet article, vous pouvez nous retrouver sur Twitter ou directement sur notre site.
Contribuer
